Good clinical practice, Clinical trial protocol, Pharmacology, Adverse event, Pre-clinical development, Informed consent
ViiV Medical Monitor, Director, ViiV R&D Physician Center of Excellence
*VIIV EMPLOYEES RECEIVE 100% OF GSK'S ROBUST BENEFITS AND TOTAL REWARDS! *POTENTIAL REMOTE OPPORTUNITY FOR THE RIGHT CANDIDATE ViiV Healthcare is ...
ViiV Medical Monitor, Director, ViiV R&D Physician Center of Excellence
*VIIV EMPLOYEES RECEIVE 100% OF GSK'S ROBUST BENEFITS AND TOTAL REWARDS! *POTENTIAL REMOTE OPPORTUNITY FOR THE RIGHT CANDIDATE ViiV Healthcare is ...
We are sorry, we could not find the related article
If you are curious about SQ1 Economy of Trust and Economy of Trust News & Trends
Please click on:
Subscribe to SQ1 Economy of Trust - Economy of Trust News & Trends
Medical/Physician Director, Clinical Development
ViiiV Healthcare is a global specialty HIV company, the only one that is 100% focused on researching and delivering new medicines for people livin...
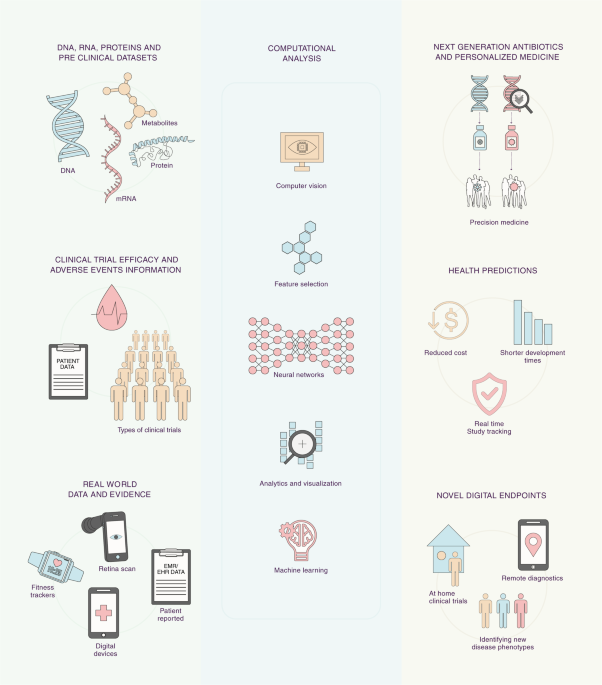
Artificial intelligence and machine learning in clinical development: a translational perspective
Future of clinical development is on the verge of a major transformation due to convergence of large new digital data sources, computing power to identify clinically meaningful patterns in ...
Patients in the driving seat
Many pharma companies talk about the importance of involving patients in trial design – but what does this really mean?
4 Life Science Trends To Pay Attention To in 2021
With COVID-19 having dominated the headlines in 2020, the coronavirus pandemic will inevitably continue to be the biggest story in 2021, but hopefully for more positive reasons. This year ...